ICTS Precision Health develops best practice guidance for investigators on the critical issues associated with genomic research.
Returning genetic results to research participants requires thoughtful planning from the research team. Precision Health provides suggestions and guidance for return of primary research results, and a guided service for returning genetic results. Browse our guidance on the Primary Findings or Secondary Findings pages.
Watch Dr. Christina Gurnett discuss return of genetic results in a lecture from the Spring 2023 Genomics in Medicine series.
ICTS Precision Health Genomic Return of Results Service
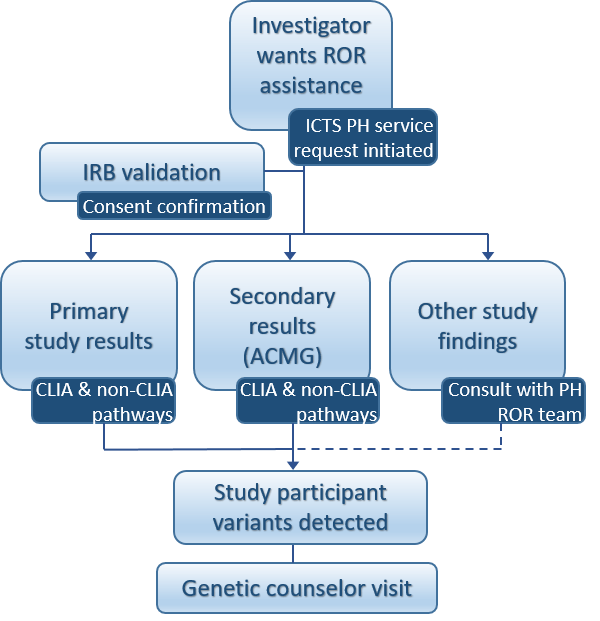
For participants who indicate on their consent form that they would like to be notified of genetic findings that may impact there health, the Investigator will return identified findings using the ICTS Precision Health Genomic Return of Results (ICTS PH gROR) Service. The service provides pathways to return primary and secondary genetic findings.
Primary study findings are defined as actionable results found based on the investigators original study approved IRB.
Secondary study findings are defined as clinically actionable genes by the American College of Medical Genetic and Genomics (ACMG).
Process for using ICTS Precision Health Genomic Return of Results Service
Step 1: Investigators complete a REDCap survey with ICTS PH ROR service.
Step 2: Investigators complete a consultation session with the precision health program scientist to discuss their study needs.
Step 3: If the investigator and program scientist are in agreement that the study can make use of the return of results service, additional REDCap surveys will be completed to gather study information. Separate pathways are available for primary study results, secondary results (ACMG) and other study findings.
Step 4: The precision health program scientist will verify with HRPO/IRB that the investigator has required consent and return of results plans approved.
Step 5: The investigator will work with the precision health return of results team. The team includes personnel with experts in sample storage, pathology and clinical genetics. Depending on their study needs, the investigator may be responsible for various costs during this process.