One Protocol One Consent
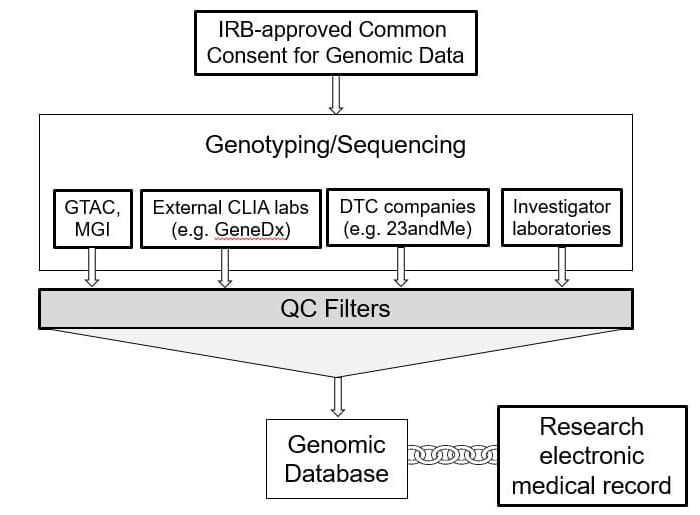
The “one protocol one consent” is an IRB approved protocol and consent that will permit the Institute of Informatics (I²) to link participant or patient genetic data that you collect to the research copy of their BJC electronic health record.
The goal of this standardized protocol and consent is to reduce the burden for individual researchers wishing to make progress in genomics and precision medicine, both for their teams and the wider research community. The standardized consent incorporates best-practices language for informing participants about genomics research and potential return of research results. It has been approved by the IRB for use by all WashU/BJH researchers and clinicians. Use of this protocol and consent as a companion to the consent for your own study or in your clinical care will facilitate future analyses for your team and lead to a large campus-wide resource for long-term research in precision medicine.
Researchers and clinicians interested in using the “one protocol one consent” should contact study coordinator Tricia Salyer at salyerp@wustl.edu to be added to the study’s protocol and provided instructions for use.
Electronic Biorepository
A partnership between BJC, St. Louis Children’s Hospital and Washington University School of Medicine has created the Electronic BioBank (BJC-WEBB). This BioBank will collect biological samples from as many people as possible who have received healthcare at Barnes-Jewish Hospital and/or Washington University School of Medicine.